Over the last ten years, the field of immunology has experienced a quiet but profound transformation. As single-cell technologies reached maturity and the cost of genetic sequencing plummeted, a new perspective emerged. Researchers stopped viewing the immune system as a mere collection of broad cell categories. Instead, they began to see it as a vibrant,
Over the last ten years, the field of immunology has experienced a quiet but profound transformation. As single-cell technologies reached maturity and the cost of genetic sequencing plummeted, a new perspective emerged. Researchers stopped viewing the immune system as a mere collection of broad cell categories. Instead, they began to see it as a vibrant, dynamic ecosystem filled with highly specialized individuals.
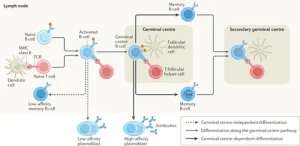
No cell type illustrates this intricate complexity better than the B cell. These cells possess an extraordinary ability to diversify, adapt, and remember past encounters. This makes them the ultimate protagonists in infection control, vaccine efficacy, and the unfortunate progression of autoimmune diseases. However, for a long time, the internal logic governing these cells remained a mystery.
In the past, scientists studied B-cells in “bulk.” They looked at large samples and hoped the average data point would tell the whole story. We now realize that this approach was often misleading. In the world of immunology, the most important biological breakthroughs often hide within the outliers—the rare cells that don’t fit the average mold.
Single-Cell BCR Profiling: A Precision Lens
Single-cell B-cell receptor (BCR) profiling has become one of the most transformative tools for decoding immune diversity. Every B-cells carries a unique receptor, which serves as its personal ID card and its primary weapon. By capturing the paired heavy- and light-chain sequences from individual cells, researchers can finally map the journey of an immune response with pinpoint accuracy.
This granular approach allows scientists to achieve several critical goals:
- Mapping Clonal Evolution: Tracking how B-cells mutate and improve their “aim” against a pathogen.
- Identifying Lineages: Pinpointing exactly which family of cells is doing the heavy lifting during an infection.
- Tracking Memory: Understanding how the body “remembers” a virus years after the initial exposure.
This level of detail is vital in our current era of rapidly shifting viral threats. Recent studies on COVID-19 and influenza show that true protection often depends on rare B-cell clones with very specific receptor features. If we only looked at the “average” cell, we would miss these elite defenders entirely.
Furthermore, oncology researchers are now using BCR profiling to study tumor-infiltrating B-cells. They want to know how these cells influence the environment surrounding a tumor. To help scientists overcome the steep technical climb of these experiments, specialized providers like Creative Biolabs have stepped in. They offer end-to-end workflows that handle everything from cell isolation to complex bioinformatic analysis. This support allows researchers to stop worrying about the plumbing of the experiment and focus entirely on the biological discovery.
The Regulatory Whisper: B-Cell miRNA Profiling
If BCR sequencing tells us the “genetic identity” of a B-cells, then microRNA (miRNA) profiling reveals its “behavioral logic.” MicroRNAs are small, non-coding RNA molecules that act as fine-tuners. They don’t build proteins themselves; instead, they control how much of a protein is made. They influence every stage of a B-cell’s life, from its initial activation to its final differentiation into a memory cell.
Experts often call miRNAs the “whisper network” of the immune system. They do not shout, but they effectively control the entire conversation. When these whispers become distorted, the results can be devastating. Recent research has identified specific miRNA signatures that act as red flags for autoimmune conditions like systemic lupus erythematosus and rheumatoid arthritis. They are also instrumental in identifying different types of B-cell lymphomas.
By utilizing miRNA profiling services, such as those provided by Creative Biolabs, scientists can quantify these tiny molecules with high resolution. This data helps link a cell’s regulatory state to its actual function in the body. When you combine this with single-cell BCR data, you get a multidimensional view of the cell. You see not only who the cell is but also what it is currently “thinking” and doing.
Building a Holistic Map: The Integrated B-Cell Atlas
The future of immunology does not belong to any single technology. Instead, it belongs to integration. The next great frontier is “multi-omics”—the practice of combining different layers of biological data into one unified picture.
Imagine a map that shows you the receptor sequence (the ID), the transcriptomics (the current activity), the epigenetics (the potential), and the miRNA profile (the regulation) of a single cell all at once. Scientists are currently working to build these comprehensive B-cell atlases. These digital maps will capture the full lifecycle of the immune response.
Why does this matter? Because a better map leads to better medicine. With a complete B-cell atlas, we can:
- Design Smarter Vaccines: We can target the exact cell lineages that provide the longest-lasting protection.
- Predict Autoimmune Flares: We can catch the “whisper” of a regulatory change before a patient feels their first symptom.
- Personalize Cancer Therapy: We can understand why some patients respond to immunotherapy while others do not.
Beyond the Technical Hurdles
The transition from bulk studies to single-cell precision is a massive leap forward, but it is also technically demanding. Analyzing thousands of individual cells generates a mountain of data. Sorting through that data requires advanced computational power and deep expertise in bioinformatics.
This is where the partnership between academia and specialized service providers becomes essential. By leveraging the infrastructure of experts like Creative Biolabs, independent labs can access high-end sequencing and analysis platforms that would otherwise be cost-prohibitive. This democratization of technology ensures that the next big breakthrough in B-cell research could come from anywhere.
We are moving away from an era of guesswork and entering an era of absolute clarity. We no longer have to settle for the “average” story. We can finally see the immune system for what it truly is: a complex, beautiful, and highly organized defense force. By decoding the BCR sequences and the miRNA whispers of individual B-cells, we are not just observing the immune system—we are learning how to speak its language.
In the coming years, this integrated approach will likely redefine our treatment of infectious diseases, cancers, and chronic inflammation. The quiet revolution in immunology is over; the era of precision B-cell medicine has officially begun. Through constant innovation and the integration of multiple data layers, we are finally unlocking the secrets of the cells that keep us safe.